All News:
Recent News
Friday, December 20th 2024
As 2024 comes to an end, we thought it a good time to reflect on a year marked by innovation in diagnostics and sample preparation and positive engagement with the world’s infectious disease community. Here are some of the highlights:

Recent News
Wednesday, August 7th 2024
Infectious disease specialist and adventurer Major Scott Pallett pitched his tent 600 km north of the Arctic Circle armed with ProtonDx Dragonfly to put the portable, rapid, multi-pathogen, molecular diagnostic platform to the test.

Recent News
Monday, February 5th 2024
As part of a planned transition aimed at strengthening our leadership with a focus on robust commercial growth in the Diagnostics and Sample Preparation industries, we are pleased to announce the appointment of Bob Enck, current Chairman and President, as CEO.

Recent News
Thurs, December 21st 2023
ProtonDx’s Regulatory Affairs Director, Dr Kasia Szostak-Lipowicz, was invited to the UN Panel on Transformative Digital Diagnostic Technologies, where she discussed how ProtonDx’s work can help bridge healthcare gaps in Africa.

Recent News
Thurs, November 2nd 2023
POCT-for-Scot Ltd. completed an evaluation of ProtonDx's multi-pathogen Dragonfly Respiratory diagnostic device. The study delves into the feasibility of using Dragonfly in Point-of-Care settings and provides insights into adoption by POC clinics!

Recent News
Thurs, October 19th 2023
Dr Rodriguez Manzano's promotion to Associate Professor in the Department of Infectious Disease at Imperial College London showcases his dedication to advancing the field of Molecular Diagnostics, with a particular focus on addressing infections and antimicrobial resistance (AMR).

Recent News
Tue, September 27th 2023
ProtonDx's Dragonfly Respiratory Panel, a portable, accurate, and rapid diagnostic device, played a pivotal role in maintaining the health and performance of UK team members throughout the Tour de France 2023.
Recent News
Tue, March 28th 2023
ProtonDx's proprietary rapid multipathogen test was a key diagnostic element used to test the Finnish cross-country skiers in The FIS Nordic World Ski Championships in Planica, Slovenia, in February - March 2023.
Company Announcements
Dragonfly Respiratory Panel Reliably Identifies Five Most Common Causes of Respiratory Infections, Joins List of UK Approved Diagnostic Products
Mon, February 6th 2023
Rapid molecular point-of-care diagnostic system reliably detects SARS-CoV-2, Flu A, Flu B, RSV and Human Rhinovirus. Clinical test results for CTDA validation achieved 100% sensitivity and 100% specificity. UK approval follows CE-IVD mark.
Company Announcements
Articles
The Respiratory Infection Season – Multiple Infections, Multiple Detection
Mon, December 12th 2022
Assistant Professor Jesus Rodriguez Manzano, Co-Founder and CSO at ProtonDx, and Deputy Director of the Centre for Antimicrobial Optimisation (CAMO) at Imperial College, shares his views on the current threat of a ‘tripledemic’ that looms over the Northern hemisphere, including many parts of Europe.
Company Announcements
ProtonDx Awarded £365,000 Defra and Innovate UK Grant to Develop Porcine Lung Disease Diagnostic
Fri, September 23rd 2022
Grant to develop automated, rapid point-of-need diagnostic to provide a sustainable and resilient revolution in detection and diagnosis of Porcine Respiratory Disease Complex (PRDC). Working in collaboration with Imperial College, farmers and industry experts to help address the global issue of rising antibiotic resistance.
Articles
Diagnostics are at the Heart of a Global AMR Solution
Wed, September 14th 2022
OPINION: Dr Jesus Rodriguez-Manzano, Co-Founder and Chief Scientific Officer (CSO) at ProtonDx, shares his thoughts on anti-microbial resistance (AMR). He discusses what we can learn from the recent COVID‑19 pandemic, including the important role that point-of-need diagnostics can play.
Company Announcements
ProtonDx Tackles Malaria Diagnosis
Fri, April 29th 2022
Lacewing, the futuristic lab-on-a-chip, diagnostic tool is one of eight initiatives promoted by Imperial College London to mark Malaria Day, 25 April, in order to demonstrate the College’s deep commitment to conquering malaria. Lacewing is being developed for health care applications by Imperial spin out company ProtonDx based at the College’s Translation and Innovation HUB (I-HUB), White City Campus.
Articles
Higher Risk for Patients with Flu and Covid-19
Mon, March 28th 2022
People infected by Covid-19 and influenza face four times the risk of requiring a ventilator and twice the risk of death. Diagnosis of which respiratory viruses are infecting the patient is crucial especially as there is no increased risk of severe illness for those with both Covid-19 and RSV or other respiratory viruses.
Articles
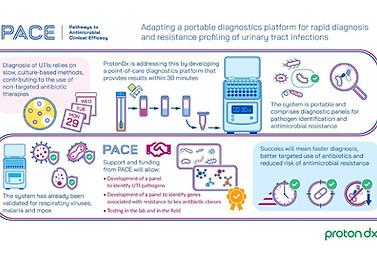
Antimicrobial Resistance Crisis Needs Better Diagnostics
Tue, February 22nd 2022
“Better, faster diagnostics will be central to tackling Antimicrobial Resistance in the future,” said ProtonDx Founder & Director, Asst Professor Jesus Rodriguez-Manzano, at a recent FORUM event organised by the Academy of Medical Sciences.





.png)








.jpg)












